Atoms

Atoms are so fundamental in the world of science, that I cannot help but to showcase them. Sounds boring and think you know all about them? Read on.
Atoms are the smallest unit of an element which still retains its properties. They are made up of protons, electrons and neutrons basically.
Atoms are the smallest unit of an element which still retains its properties. They are made up of protons, electrons and neutrons basically.
The Atomic Model
http://education.jlab.org/qa/atom_model_02.gif
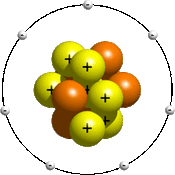
Back to basics. In here, the balls marked with a plus are the protons, which have positive charges. The red balls are neutrons, with a neutral charge. The balls marked with a minus are the electrons, which have a negative charge. Each shell can hold a limited number of electrons. For all elements, 1st and 2nd shell can hold 2 electrons and 8 electrons respectively. After the first 20 elements in the periodic table (after Calcium), which can hold a maximum of 8 electrons in the third shell, the third shell can hold up to 18 electrons.
Some terms
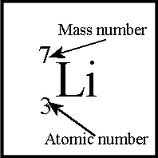
Atomic number is the number of protons
Mass number is the number of protons and the number of neutrons combined
In this figure, the number of neutrons can be deduced to be 4, as there are 3 atoms and 7 atoms+neutrons in Lithium.
Some terms
Atomic number is the number of protons
Mass number is the number of protons and the number of neutrons combined
In this figure, the number of neutrons can be deduced to be 4, as there are 3 atoms and 7 atoms+neutrons in Lithium.
Here's the interesting part...
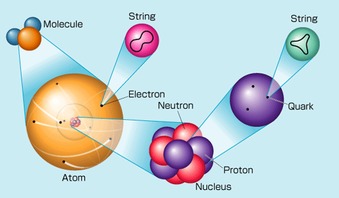
There are actually smaller particles which reside in the atom. All these smaller particles (including protons, electrons and neutrons), are called subatomic particles. Some examples would be: neutrinos, the Higgs boson, leptons etc.
But let me focus on a type of elementary particle- quarks. What are elementary particles, you may ask. In layman's terms, it means that they are the building blocks which make up matter. Quarks are the smallest particles known to man today, and the odd thing is, they cannot be seen! They are only detected when their actions cause effects, like colliding into another particle.
There are six types of quarks: Up, Down, Charm, Strange, Top and Bottom. Why are quarks... quarks? Murray Gell-Mann, and American physicist, read the line "Three quarks for Muster Mark..." in a book "Finnegan's Wake" and thus named these elementary particles: QUARKS. Funny names?
However, to go in-depth to find out what each specifically does, we would be diving into the world of particle physics, which would mean we are digressing from chemistry by a lot... Still, Science is meant to allow multiple disciplines to be studied together, just like how I am trying to pave a connection between chemistry right now!
But let me focus on a type of elementary particle- quarks. What are elementary particles, you may ask. In layman's terms, it means that they are the building blocks which make up matter. Quarks are the smallest particles known to man today, and the odd thing is, they cannot be seen! They are only detected when their actions cause effects, like colliding into another particle.
There are six types of quarks: Up, Down, Charm, Strange, Top and Bottom. Why are quarks... quarks? Murray Gell-Mann, and American physicist, read the line "Three quarks for Muster Mark..." in a book "Finnegan's Wake" and thus named these elementary particles: QUARKS. Funny names?
However, to go in-depth to find out what each specifically does, we would be diving into the world of particle physics, which would mean we are digressing from chemistry by a lot... Still, Science is meant to allow multiple disciplines to be studied together, just like how I am trying to pave a connection between chemistry right now!
Enjoy a video on particle physics that explains simply the complicated world of particles.
Isotopes
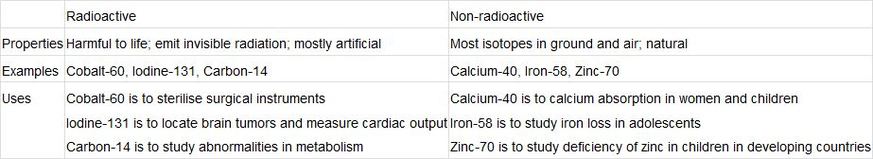
Neutrons are pretty interesting, too. Generally, isotopes are atoms with the same atomic number (number of atoms) but different mass number (number of neutrons).
What is different? Well, Isotopes have different physical properties as they depend on the masses of atoms/molecules. Then you may ask, why bother to have different isotopes? Categorise them into two groups first.
What is different? Well, Isotopes have different physical properties as they depend on the masses of atoms/molecules. Then you may ask, why bother to have different isotopes? Categorise them into two groups first.
Frederick Soddy, an English radiochemist, first introduced the idea of isotopes while trying to explain radioactivity. It was Joseph John Thomson, a British physicist, who discovered the first isotope (when he found that neon had more than one positive ion; thus naming them isotopes).
All this said, let's end off with a video that showcases a way to produce isotpes in medicine
All this said, let's end off with a video that showcases a way to produce isotpes in medicine