Johann Dobereiner’s and Johann Newlands’ attempts to classify the elements
ACE
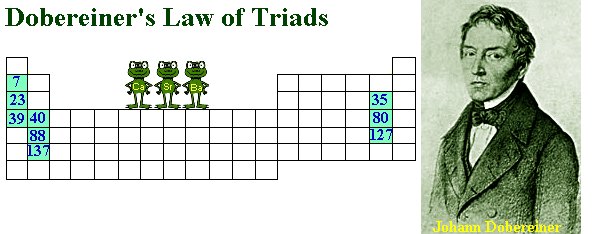
Johann Wolfgang Dobereiner, a German scientist, was the first person to classify the elements into groups based on John Dalton, a fellow scientist’s suggestions. After trying since 1817, elements with similar properties were grouped into clusters of three called ‘Triads’. The atomic mass of the middle element in the triad was the special feature. That mass was the approximate arithmetic mean of the other two elements in the triad when the elements were arranged in order of their increasing atomic mass. Here is an example:
Ca Sr Ba (40 + 137) ÷ 2 = 88
40 88 137
Taken from //http://web.fccj.org/~ethall/period/period.htm//
In 1829, Dobereiner proposed this finding to become the Law of Triads. However, there were loopholes in his “law”. For one, a large number of similar elements could not be grouped into triads, like iron, manganese, nickel, cobalt, zinc and copper. Another thing is that Dobereiner could only identify three triads successfully from the elements known at that time, so there was little supporting evidence.
Johann Wolfgang Dobereiner, a German scientist, was the first person to classify the elements into groups based on John Dalton, a fellow scientist’s suggestions. After trying since 1817, elements with similar properties were grouped into clusters of three called ‘Triads’. The atomic mass of the middle element in the triad was the special feature. That mass was the approximate arithmetic mean of the other two elements in the triad when the elements were arranged in order of their increasing atomic mass. Here is an example:
Ca Sr Ba (40 + 137) ÷ 2 = 88
40 88 137
Taken from //http://web.fccj.org/~ethall/period/period.htm//
In 1829, Dobereiner proposed this finding to become the Law of Triads. However, there were loopholes in his “law”. For one, a large number of similar elements could not be grouped into triads, like iron, manganese, nickel, cobalt, zinc and copper. Another thing is that Dobereiner could only identify three triads successfully from the elements known at that time, so there was little supporting evidence.
Dobereiner's work focused on the relationships among the elements in a triad but could not find any relationship linking the triads. John Alexander Reina Newlands, aka Johann Newlands, was an English chemist who took the next step towards building the periodic table. He classified the 56 known at the time into eleven groups based on similar physical properties. He was the first to arrange elements in order of the relative atomic weights. Through this method, he found that each element was similar to the element eight places further on. This was called “The Law of Octaves” In Newlands’ periodic table, periods were going down and groups were going across the table. It was the opposite from the modern periodic table.
His work also had loopholes. It was discovered that after repeating his table regularly, some elements which had no relationship with other ones were found to be in sequence. He had failed to provide space for the discovery of additional elements at that time where elements were occasionally being discovered. Still, his incomplete periodic table in 1864 led to his prediction of germanium. His mistakes were probably caused by his enthrallment of trying to force the elements into the pattern of a music scale, in which one note in a key is separated from its octave by an interval of seven notes.
His work also had loopholes. It was discovered that after repeating his table regularly, some elements which had no relationship with other ones were found to be in sequence. He had failed to provide space for the discovery of additional elements at that time where elements were occasionally being discovered. Still, his incomplete periodic table in 1864 led to his prediction of germanium. His mistakes were probably caused by his enthrallment of trying to force the elements into the pattern of a music scale, in which one note in a key is separated from its octave by an interval of seven notes.
Nevertheless, these two scientists have made a great contribution to the modern periodic table. Without them, our understanding of the elements might not be so profound today. They had devised clever ways to group the elements so that scientists of today can further develop their work and learn from their mistakes and achievements. I'm sure they deserve our utmost respect for spearheading such a massive journey in chemistry.